At the time of writing this article, an ounce of platinum costs $992, while a kilogram is priced at $32,000.
That is a pretty hefty price for a metal, so what makes platinum so expensive? What drives its value to such high levels?
Why Platinum is so expensive and valuable
Rarity
Platinum is 10 times rarer than gold, with only about 200-250 tons of platinum mined every year, compared to some 3,000 tons of gold.
70-80% of the platinum produced every year comes from South Africa, some 10-20% is mined in Russia and the rest being sourced from North & South America.
However, the places with the most easily accessible platinum deposits have already been mined.
As a result, platinum miners must now dig deeper and deeper in order to find the precious metal, which adds to the production and environmental cost of platinum.
With old platinum deposits now depleted, and no new major ones found, the yearly production of platinum has reached a plateau and is unlikely to grow foreseeable future.
Platinum jewelry is denser than gold & harder to make
Despite platinum being cheaper than gold ($992 per ounce vs $1750 per ounce), jewelry made from platinum is usually more expensive than gold alternatives.
This is because platinum is denser than gold, meaning there are more grams of platinum in a wedding ring, compared to an identical ring made out of gold.
On top of that, because of its chemical properties platinum can be manufactured into jewelry at a much higher purity compared to gold.
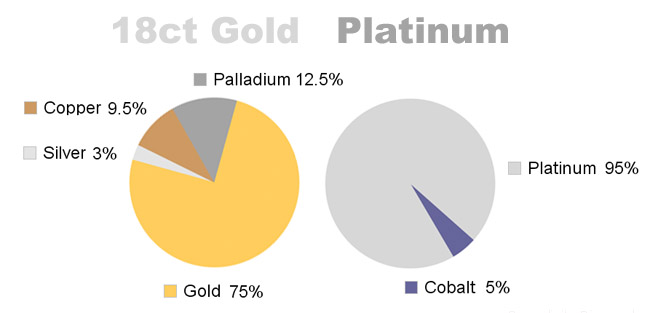
For example, a typical platinum wedding ring has a 90-95% purity, meaning 90%+ of the ring is platinum, with the rest being various alloy materials.
By comparison, a typical 14 karat white gold wedding ring is 59% gold, with the remaining 41% being various alloys such as palladium or nickel.
An 18 karat wedding ring is 75% gold and 25% various other alloys.
Platinum is more difficult to turn into jewelry compared to gold.
Platinum is a harder metal to turn into jewelry compared to 14/18k gold and requires its own specialized skill set.
Because most people gravitate to gold jewelry instead of platinum, most jewelers rarely work with platinum and don’t have the skills required to handle the metal.
As a result, jewelers that are skilled in working with platinum are relatively few, so they command a premium for their services.
Platinum has many different applications
Most of the reasons platinum is in high demand is because it offers a lot of practical uses that few, or no other metal can accomplish.
For example, nearly half of cancer patients use drugs that contain platinum, such as cisplatin. Platinum is a useful element in medicine because it isn’t toxic to humans and is non-reactive, making it compatible with human tissues.
Around 45% of all platinum produced every year is used to manufacture catalytic converters. These are car components responsible for reducing the toxicity and amount of emissions produced by cars.
Some 20% is used to manufacture drugs and medical equipment, hard drives, chemical and petroleum refining, glass making equipment, oxygen sensors, turbine engines and more.
Only 30-35% of all platinum produced worldwide is used to make jewelry.
High demand
Just like everything else in an economy, platinum is also subject to the invisible hand of supply and demand.
When analyzing the long of list of practical applications of platinum, its capacity to store wealth and overall rarity, the price of platinum begins to make sense.
Interestingly though, platinum is cheaper than gold: $992 per ounce compared to $1750.
This is despite the fact that we produce just 200-250 tons of platinum per year, compared to 3000 tons of gold.
On top of this, gold has been in production for thousands of years. By current estimates, there are around 150,000 to 250,000 tons of gold currently in circulation.
By comparison, most estimates claim the total amount of platinum we’ve produced is around 5,000 to 10,000 tons, so there’s around 15-50 times less platinum in circulation than gold.
Taking this into account, it’s safe to say that if platinum was as common as gold, supply and demand would diminish its price to be closer to that of silver than of gold.
Platinum is a safe haven investment
Like gold and silver, platinum is a safe investment instrument people use to protect their money against inflation or economic downturns.
The reason gold, silver and platinum are used to store value is partly due to the rarity of these metals, but most importantly it’s because they do not corrode.
Iron for example is terrible as a means to store of value because it is abundant, but also because it rusts away into nothing given enough time.
When stored in the right conditions, a gold or platinum ingot can last for decades without any noticeable degradation. This gives investors peace of mind, since they know can resell the metal at market value at any point in the future.
This phenomenon of using gold and silver as a means to store wealth is ancient, and was independently discovered by nearly every major human society.
While it doesn’t have the same history behind it, platinum has now joined silver and gold as a means of storing wealth.
Difficult manufacturing
Throughout history, gold and silver were the two precious metals that dominated trade and used as stores of value.
One of the biggest reason for their success is the fact that both gold and silver are easy to mine, and simple to isolate in relatively pure, concentrated forms through the use of fire and melting.
Platinum, however, is a much more difficult metal to work with.
The technology required to efficiently process platinum didn’t exist until 100-150 years ago which explains why it wasn’t a widely used metal in the early human history.
For starters, you need around 12 tons of ore to produce a single ounce of platinum.
Turning this ore into platinum is a complex process that involves:
- Crushing the ore into small particles.
- Mixing the crushed ore with water and other chemical reagents.
- Blowing air into the mixture to force platinum particles to the top of this chemical soup.
- Collect the froth at the top of the soup, then dry it into a powder.
- Heat the dry powder to extremely high temperatures to remove impurities.
- Apply other chemical processes to further purify the powder, removing any sulfur, iron, nickel etc.
- Dissolve the remaining concentrate with aqua regia (3/4 hydrochloric acid & 1/4 nitric acid).
- Finalize the purification process and turn the remaining mineral concentrate into platinum.
This is just the production stage of platinum. Jewelers looking to turn platinum into rings, necklaces etc. have to apply their own refining process.
This too is much more complex than the one used for creating gold and silver jewelry, which adds to the cost of the end product.